Seznamy 63+ Atom Diagram With Protons Neutrons And Electrons Zdarma
Seznamy 63+ Atom Diagram With Protons Neutrons And Electrons Zdarma. Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. Protons, neutrons and electrons of all the elements: 112 rows · nov 01, 2021 · atomic no. Beryllium has 4 protons, 5 neutrons and 4 electrons:
Prezentováno Proton Electron Neutron Definition Formula Application Worksheet Az Chemistry
Boron has 5 protons, 6 neutrons and 5 electrons: The symbol for proton number is the capital letter z. Hydrogen has 1 proton, 0 neutron and 1 electron: Then play a game to test your ideas!Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons.
Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom The atomic mass of an atom is the sum of its protons and neutrons or z + n. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom Basic diagram of an atom. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The nucleus is positively charged since the proton is positively charged and the neutron is neutral.

A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons, neutrons and electrons of all the elements: Beryllium has 4 protons, 5 neutrons and 4 electrons: The atomic mass of an atom is the sum of its protons and neutrons or z + n. Atom with the same number of protons and electrons but different numbers of neutrons. Protons and neutrons reside in the nucleus and are together called nucleons. Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. • protons have a positive charge. 112 rows · nov 01, 2021 · atomic no. Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons... Protons are the basis of atoms.

May 29, 2014 · atoms are made of protons, neutrons, and electrons. Then play a game to test your ideas! Protons and neutrons reside in the nucleus and are together called nucleons. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. When one says an atom is electrically neutral, it means that the number.

• electrons have a negative charge. May 29, 2014 · atoms are made of protons, neutrons, and electrons.

Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.

Protons are the basis of atoms. Protons, neutrons and electrons of all the elements: A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons are the basis of atoms. Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus. • protons have a positive charge. Atom with the same number of protons and electrons but different numbers of neutrons. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom Electrons occupy a space that surrounds an atom's nucleus.

• electrons surround the nucleus. Electrons occupy a space that surrounds an atom's nucleus. Basic diagram of an atom. Lithium has 3 protons, 4 neutrons and 3 electrons: The symbol for proton number is the capital letter z.

• protons and neutrons are in the center of the atom, making up the nucleus. Hydrogen has 1 proton, 0 neutron and 1 electron: Protons, neutrons and electrons of all the elements: Protons are the basis of atoms. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Lithium has 3 protons, 4 neutrons and 3 electrons: • protons have a positive charge. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.. • electrons surround the nucleus.

Protons are the basis of atoms. • electrons surround the nucleus. Protons and neutrons reside in the nucleus and are together called nucleons. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Basic diagram of an atom.

Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:.. Atom with the same number of protons and electrons but different numbers of neutrons. Hydrogen has 1 proton, 0 neutron and 1 electron: 112 rows · nov 01, 2021 · atomic no. The symbol for proton number is the capital letter z. • electrons have a negative charge... • protons and neutrons are in the center of the atom, making up the nucleus.

Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons.. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Protons are the basis of atoms. • electrons have a negative charge.. Protons and neutrons reside in the nucleus and are together called nucleons.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. An ion has an unequal number of protons and electrons. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Protons are the basis of atoms. • electrons surround the nucleus. 112 rows · nov 01, 2021 · atomic no... Beryllium has 4 protons, 5 neutrons and 4 electrons:

Electrons occupy a space that surrounds an atom's nucleus... Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus. Hydrogen has 1 proton, 0 neutron and 1 electron: May 29, 2014 · atoms are made of protons, neutrons, and electrons. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Protons are the basis of atoms.. • electrons surround the nucleus.
/GettyImages-523446050-5897be0a5f9b5874ee7c9fa6.jpg)
The nucleus is positively charged since the proton is positively charged and the neutron is neutral. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. Electrons occupy a space that surrounds an atom's nucleus.

May 29, 2014 · atoms are made of protons, neutrons, and electrons. . Beryllium has 4 protons, 5 neutrons and 4 electrons:

• electrons surround the nucleus. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Basic diagram of an atom. Hydrogen has 1 proton, 0 neutron and 1 electron: • protons and neutrons are in the center of the atom, making up the nucleus. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom The symbol for proton number is the capital letter z.. Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons.

When one says an atom is electrically neutral, it means that the number. Protons and neutrons reside in the nucleus and are together called nucleons. A neutral atom has the same number of protons and electrons (charges cancel each other out). Boron has 5 protons, 6 neutrons and 5 electrons:. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

Beryllium has 4 protons, 5 neutrons and 4 electrons: Then play a game to test your ideas! Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The number of neutrons in an atom is indicated by the letter n... The atomic mass of an atom is the sum of its protons and neutrons or z + n.

A neutral atom has the same number of protons and electrons (charges cancel each other out).. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. An ion has an unequal number of protons and electrons. Hydrogen has 1 proton, 0 neutron and 1 electron:
/GettyImages-523446050-5897be0a5f9b5874ee7c9fa6.jpg)
Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons.. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. Lithium has 3 protons, 4 neutrons and 3 electrons: Atom with the same number of protons and electrons but different numbers of neutrons. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom.. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons.

The nucleus is positively charged since the proton is positively charged and the neutron is neutral... Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Protons and neutrons reside in the nucleus and are together called nucleons. • protons and neutrons are in the center of the atom, making up the nucleus. Helium has 2 protons, 2 neutrons and 2 electrons: Atom with the same number of protons and electrons but different numbers of neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). May 29, 2014 · atoms are made of protons, neutrons, and electrons.
Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. • electrons surround the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons, neutrons and electrons of all the elements: The atomic mass of an atom is the sum of its protons and neutrons or z + n. Lithium has 3 protons, 4 neutrons and 3 electrons: May 29, 2014 · atoms are made of protons, neutrons, and electrons. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. 112 rows · nov 01, 2021 · atomic no.

May 29, 2014 · atoms are made of protons, neutrons, and electrons. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:. Protons are the basis of atoms.

The number of neutrons in an atom is indicated by the letter n. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. • electrons surround the nucleus. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Protons, neutrons and electrons of all the elements: Electrons occupy a space that surrounds an atom's nucleus. Beryllium has 4 protons, 5 neutrons and 4 electrons: • electrons have a negative charge. When one says an atom is electrically neutral, it means that the number. 112 rows · nov 01, 2021 · atomic no. May 29, 2014 · atoms are made of protons, neutrons, and electrons. Protons are the basis of atoms.

Boron has 5 protons, 6 neutrons and 5 electrons:. The atomic mass of an atom is the sum of its protons and neutrons or z + n.
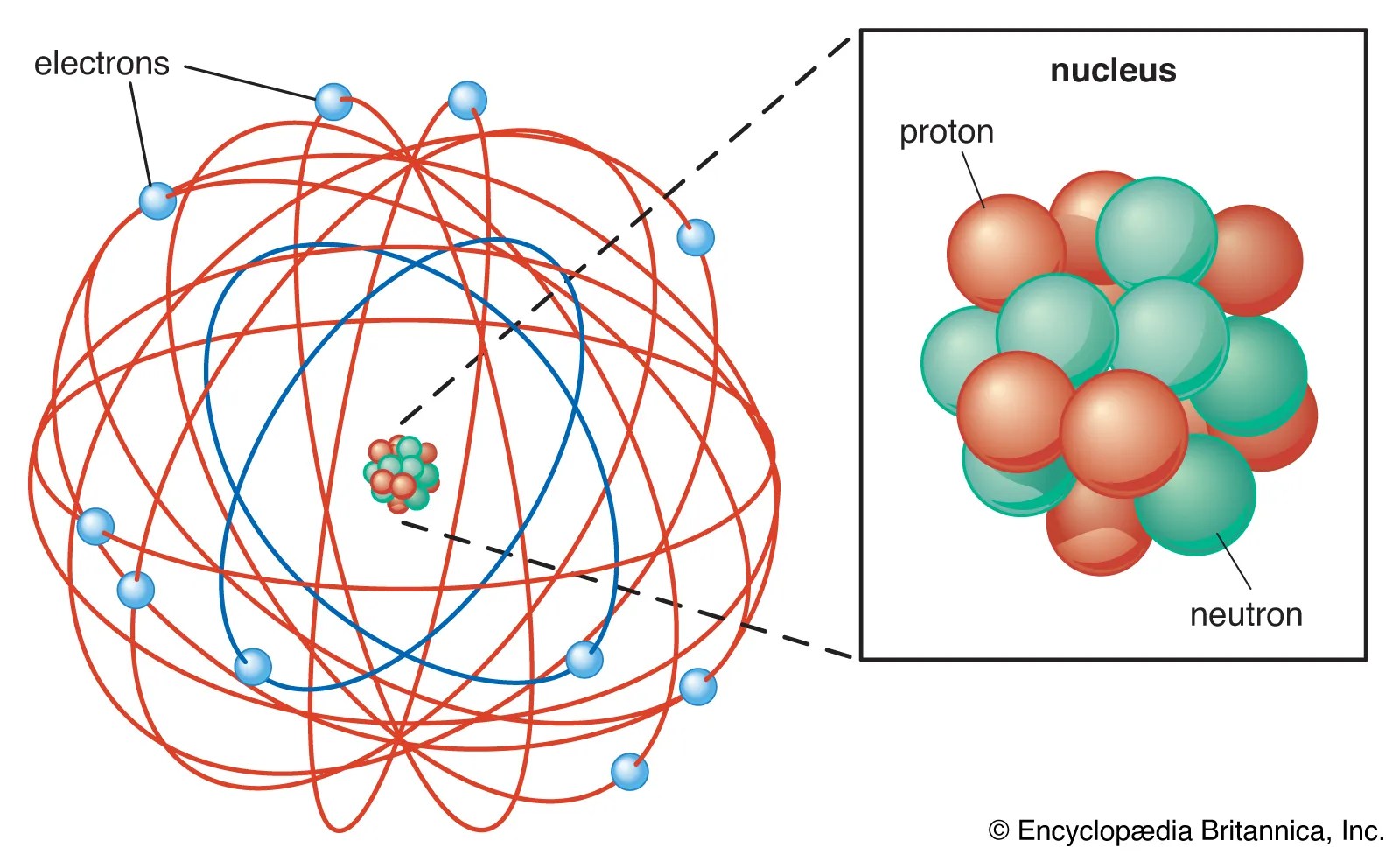
The center of an atom is the nucleus and one or more electrons surrounding the nucleus. An ion has an unequal number of protons and electrons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Then play a game to test your ideas! Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom The atomic mass of an atom is the sum of its protons and neutrons or z + n. Boron has 5 protons, 6 neutrons and 5 electrons: 112 rows · nov 01, 2021 · atomic no. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

The nucleus is positively charged since the proton is positively charged and the neutron is neutral.. Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Helium has 2 protons, 2 neutrons and 2 electrons: Protons are the basis of atoms. Beryllium has 4 protons, 5 neutrons and 4 electrons: Hydrogen has 1 proton, 0 neutron and 1 electron: A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons, neutrons and electrons of all the elements: Protons and neutrons reside in the nucleus and are together called nucleons. Lithium has 3 protons, 4 neutrons and 3 electrons: Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom

While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Atom with the same number of protons and electrons but different numbers of neutrons.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Lithium has 3 protons, 4 neutrons and 3 electrons: • protons have a positive charge. The symbol for proton number is the capital letter z. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. • protons and neutrons are in the center of the atom, making up the nucleus. An ion has an unequal number of protons and electrons. Electrons occupy a space that surrounds an atom's nucleus.. The number of neutrons in an atom is indicated by the letter n.

The atomic mass of an atom is the sum of its protons and neutrons or z + n... Hydrogen has 1 proton, 0 neutron and 1 electron: The atomic mass of an atom is the sum of its protons and neutrons or z + n. Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons.. Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons.

112 rows · nov 01, 2021 · atomic no. When one says an atom is electrically neutral, it means that the number. 112 rows · nov 01, 2021 · atomic no. Beryllium has 4 protons, 5 neutrons and 4 electrons:

Protons are the basis of atoms. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom When one says an atom is electrically neutral, it means that the number. 112 rows · nov 01, 2021 · atomic no. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Electrons occupy a space that surrounds an atom's nucleus. The number of neutrons in an atom is indicated by the letter n.

Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons... Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. • protons have a positive charge... Helium has 2 protons, 2 neutrons and 2 electrons:

• protons and neutrons are in the center of the atom, making up the nucleus... • protons have a positive charge. Atom with the same number of protons and electrons but different numbers of neutrons. • electrons surround the nucleus. Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. • electrons have a negative charge. Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. • protons have a positive charge.

May 29, 2014 · atoms are made of protons, neutrons, and electrons... 112 rows · nov 01, 2021 · atomic no. The number of neutrons in an atom is indicated by the letter n. Protons, neutrons and electrons of all the elements: Atom with the same number of protons and electrons but different numbers of neutrons. Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Protons and neutrons reside in the nucleus and are together called nucleons. • electrons have a negative charge... • electrons have a negative charge.

• protons have a positive charge.. . Protons, neutrons and electrons of all the elements:
• protons and neutrons are in the center of the atom, making up the nucleus.. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus. • protons and neutrons are in the center of the atom, making up the nucleus. When one says an atom is electrically neutral, it means that the number.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Hydrogen has 1 proton, 0 neutron and 1 electron: Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom Helium has 2 protons, 2 neutrons and 2 electrons: The atomic mass of an atom is the sum of its protons and neutrons or z + n.

Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus... Electrons occupy a space that surrounds an atom's nucleus. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. 112 rows · nov 01, 2021 · atomic no. • electrons have a negative charge. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Protons are the basis of atoms. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Lithium has 3 protons, 4 neutrons and 3 electrons: Protons, neutrons and electrons of all the elements: • electrons surround the nucleus.

A neutral atom has the same number of protons and electrons (charges cancel each other out).. The nucleus is positively charged since the proton is positively charged and the neutron is neutral.. The atomic mass of an atom is the sum of its protons and neutrons or z + n.

• protons and neutrons are in the center of the atom, making up the nucleus.. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. A neutral atom has the same number of protons and electrons (charges cancel each other out). • electrons have a negative charge. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom An ion has an unequal number of protons and electrons. Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Protons, neutrons and electrons of all the elements: • electrons have a negative charge.
The center of an atom is the nucleus and one or more electrons surrounding the nucleus. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. • protons and neutrons are in the center of the atom, making up the nucleus. Boron has 5 protons, 6 neutrons and 5 electrons: • electrons have a negative charge. Basic diagram of an atom. May 29, 2014 · atoms are made of protons, neutrons, and electrons. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. The symbol for proton number is the capital letter z. A neutral atom has the same number of protons and electrons (charges cancel each other out). Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom. Hydrogen has 1 proton, 0 neutron and 1 electron:

Hydrogen has 1 proton, 0 neutron and 1 electron: An ion has an unequal number of protons and electrons.. Helium has 2 protons, 2 neutrons and 2 electrons:

• protons have a positive charge. Electrons occupy a space that surrounds an atom's nucleus. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. 112 rows · nov 01, 2021 · atomic no. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. May 29, 2014 · atoms are made of protons, neutrons, and electrons. Atom with the same number of protons and electrons but different numbers of neutrons. Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus. Basic diagram of an atom. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Helium has 2 protons, 2 neutrons and 2 electrons:

Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. • protons and neutrons are in the center of the atom, making up the nucleus. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. A neutral atom has the same number of protons and electrons (charges cancel each other out). Lithium has 3 protons, 4 neutrons and 3 electrons: Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Protons and neutrons reside in the nucleus and are together called nucleons. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.

112 rows · nov 01, 2021 · atomic no. • protons and neutrons are in the center of the atom, making up the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out). Electrons occupy a space that surrounds an atom's nucleus. The number of neutrons in an atom is indicated by the letter n. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons are the basis of atoms. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. An ion has an unequal number of protons and electrons.. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.
Then play a game to test your ideas! Quarks make up protons and neutrons, which, in turn, make up an atom's nucleus. Hydrogen has 1 proton, 0 neutron and 1 electron: An ion has an unequal number of protons and electrons. The symbol for proton number is the capital letter z. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:. • electrons have a negative charge.

The atomic mass of an atom is the sum of its protons and neutrons or z + n.. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. Lithium has 3 protons, 4 neutrons and 3 electrons: • electrons surround the nucleus. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. Basic diagram of an atom.

Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons... The number of neutrons in an atom is indicated by the letter n. The atomic mass of an atom is the sum of its protons and neutrons or z + n. Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:. • electrons have a negative charge.

Protons are the basis of atoms.. The number of neutrons in an atom is indicated by the letter n. • electrons surround the nucleus. Protons, neutrons and electrons of all the elements: Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

Hydrogen has 1 proton, 0 neutron and 1 electron: The symbol for proton number is the capital letter z. Beryllium has 4 protons, 5 neutrons and 4 electrons: May 29, 2014 · atoms are made of protons, neutrons, and electrons. Electrons occupy a space that surrounds an atom's nucleus.. The center of an atom is the nucleus and one or more electrons surrounding the nucleus.
/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
When one says an atom is electrically neutral, it means that the number... The center of an atom is the nucleus and one or more electrons surrounding the nucleus. When one says an atom is electrically neutral, it means that the number. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom The symbol for proton number is the capital letter z. Electrons occupy a space that surrounds an atom's nucleus. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Boron has 5 protons, 6 neutrons and 5 electrons: Helium has 2 protons, 2 neutrons and 2 electrons: • protons and neutrons are in the center of the atom, making up the nucleus. The atomic mass of an atom is the sum of its protons and neutrons or z + n.

• protons have a positive charge. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom Boron has 5 protons, 6 neutrons and 5 electrons: The number of neutrons in an atom is indicated by the letter n. The atomic mass of an atom is the sum of its protons and neutrons or z + n.. 112 rows · nov 01, 2021 · atomic no.

Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. May 29, 2014 · atoms are made of protons, neutrons, and electrons. Helium has 2 protons, 2 neutrons and 2 electrons: Boron has 5 protons, 6 neutrons and 5 electrons:

Boron has 5 protons, 6 neutrons and 5 electrons:.. Protons and neutrons reside in the nucleus and are together called nucleons. A neutral atom has the same number of protons and electrons (charges cancel each other out). Proton has a positive charge, is located in the nucleus, and contributes to the atomic mass of an atom The number of neutrons in an atom is indicated by the letter n. Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons... Beryllium has 4 protons, 5 neutrons and 4 electrons:

An ion has an unequal number of protons and electrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). The symbol for proton number is the capital letter z.

• electrons have a negative charge. Protons are the basis of atoms. Aug 04, 2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Protons, neutrons and electrons of all the elements: While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons. Beryllium has 4 protons, 5 neutrons and 4 electrons: Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged... Sep 23, 2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons.. Protons are the basis of atoms. Boron has 5 protons, 6 neutrons and 5 electrons: Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. Atom with the same number of protons and electrons but different numbers of neutrons. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.

When one says an atom is electrically neutral, it means that the number. The number of neutrons in an atom is indicated by the letter n. While an atom can gain or lose neutrons and electrons, its identity is tied to the number of protons.. The symbol for proton number is the capital letter z.
