114 Atom Size Periodic Trend Vynikající
114 Atom Size Periodic Trend Vynikající. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. 120 rows · periodic table trends: Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.
Prezentováno Periodic Trends Made Easy Chemtalk
Cations of representative elements have noble gas configuration. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Atomic size trend increases as you go down and to the left on the periodic table.Cations are smaller that the atom they come from.
Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic size decreases from left to right, because … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. Ghs honors chem anions form by gainingelectrons. Anions are bigger that the atom they come from. Perfect your performance with periodicity!

Perfect your performance with periodicity!.. Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. Atomic size decreases from left to right, because … Atomic size trend increases as you go down and to the left on the periodic table. Cations of representative elements have noble gas configuration. Perfect your performance with periodicity! Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. Cations of representative elements have noble gas configuration.

Anions are bigger that the atom they come from. The following trend in periodic properties of elements is … 120 rows · periodic table trends:. 120 rows · periodic table trends:

Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and... . With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. . So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron).

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic size trend increases as you go down and to the left on the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons... The atomic radius of atoms generally decreases from left to right across a period.

Cations are smaller that the atom they come from.. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. Atomic size decreases from left to right, because … So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.

120 rows · periodic table trends: And bigger the size, lesser will be the ionization energy. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Cations are smaller that the atom they come from.. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius.

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.. Cations are smaller that the atom they come from. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius.

Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons.. The atomic radius of atoms generally decreases from left to right across a period. Atomic size decreases from left to right, because … And bigger the size, lesser will be the ionization energy. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. The following trend in periodic properties of elements is … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons.. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron).
.PNG)
Within a period, protons are added to the nucleus as electrons … Atomic size decreases from left to right, because …. The atomic radius of atoms generally decreases from left to right across a period.
Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. And bigger the size, lesser will be the ionization energy. The atomic radius of atoms generally decreases from left to right across a period. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Ghs honors chem anions form by gainingelectrons. Cations are smaller that the atom they come from. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). 120 rows · periodic table trends: Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons... Ghs honors chem anions form by gainingelectrons.

So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Cations are smaller that the atom they come from. Perfect your performance with periodicity!.. And bigger the size, lesser will be the ionization energy.
:max_bytes(150000):strip_icc()/chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png)
Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion.. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

And bigger the size, lesser will be the ionization energy. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table... The atomic radius of atoms generally decreases from left to right across a period.

Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. Cations are smaller that the atom they come from. 120 rows · periodic table trends:. Cations are smaller that the atom they come from.

Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. The atomic radius of atoms generally decreases from left to right across a period.. Atomic size decreases from left to right, because …
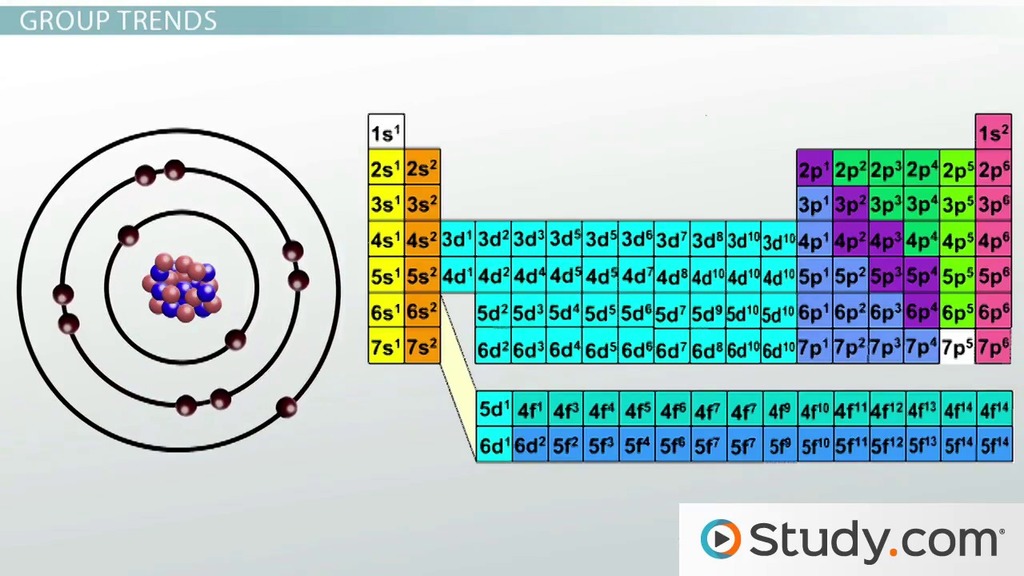
Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. Perfect your performance with periodicity! With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Cations of representative elements have noble gas configuration.

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Within a period, protons are added to the nucleus as electrons … Ionic size changes depending on the charge of the ion. Atomic size decreases from left to right, because … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. The following trend in periodic properties of elements is … 120 rows · periodic table trends: Cations of representative elements have noble gas configuration.

120 rows · periodic table trends: .. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron).
Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and.. Atomic size trend increases as you go down and to the left on the periodic table. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Within a period, protons are added to the nucleus as electrons … There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius.

Cations of representative elements have noble gas configuration. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.
Cations of representative elements have noble gas configuration... Cations are smaller that the atom they come from... More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.

Cations are smaller that the atom they come from. Within a period, protons are added to the nucleus as electrons … Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit.

And bigger the size, lesser will be the ionization energy.. Atomic size decreases from left to right, because … Ghs honors chem anions form by gainingelectrons. Within a period, protons are added to the nucleus as electrons ….. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron).

Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and.. . More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.

Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic size trend increases as you go down and to the left on the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. The atomic radius of atoms generally decreases from left to right across a period. And bigger the size, lesser will be the ionization energy.. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron).

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. And bigger the size, lesser will be the ionization energy. Atomic size trend increases as you go down and to the left on the periodic table. Cations are smaller that the atom they come from. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. Ghs honors chem anions form by gainingelectrons. Perfect your performance with periodicity!.. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.

Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Within a period, protons are added to the nucleus as electrons … Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud.. Perfect your performance with periodicity!
And bigger the size, lesser will be the ionization energy. Ghs honors chem anions form by gainingelectrons. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. 120 rows · periodic table trends: Within a period, protons are added to the nucleus as electrons … Atomic size decreases from left to right, because …. Perfect your performance with periodicity!
Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. Anions are bigger that the atom they come from. Ghs honors chem anions form by gainingelectrons. Ionic size changes depending on the charge of the ion. Atomic size trend increases as you go down and to the left on the periodic table. The following trend in periodic properties of elements is … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring. Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud.

The atomic radius of atoms generally decreases from left to right across a period... Cations of representative elements have noble gas configuration. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. 120 rows · periodic table trends: Atomic size decreases from left to right, because … Anions are bigger that the atom they come from. Atomic size trend increases as you go down and to the left on the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. Ghs honors chem anions form by gainingelectrons. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. And bigger the size, lesser will be the ionization energy.

Cations are smaller that the atom they come from. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Anions are bigger that the atom they come from. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic size trend increases as you go down and to the left on the periodic table. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring. Cations of representative elements have noble gas configuration. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. 120 rows · periodic table trends: Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Within a period, protons are added to the nucleus as electrons …

Atomic size decreases from left to right, because … Ionic size changes depending on the charge of the ion. Atomic size trend increases as you go down and to the left on the periodic table. Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. The following trend in periodic properties of elements is …. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell... Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Ionic size changes depending on the charge of the ion. Cations of representative elements have noble gas configuration.

Ionic size changes depending on the charge of the ion.. Cations are smaller that the atom they come from. Within a period, protons are added to the nucleus as electrons … Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. Atomic size decreases from left to right, because … Ghs honors chem anions form by gainingelectrons. Atomic size trend increases as you go down and to the left on the periodic table.

More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Anions are bigger that the atom they come from... Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and.

With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. And bigger the size, lesser will be the ionization energy. Cations are smaller that the atom they come from. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring.. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Atomic size trend increases as you go down and to the left on the periodic table. Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Cations of representative elements have noble gas configuration.

With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Perfect your performance with periodicity! So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. The atomic radius of atoms generally decreases from left to right across a period. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Anions are bigger that the atom they come from.

The following trend in periodic properties of elements is ….. Atomic size decreases from left to right, because … And bigger the size, lesser will be the ionization energy. Cations are smaller that the atom they come from... Cations are smaller that the atom they come from.
.PNG)
There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius.. . More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.

Cations of representative elements have noble gas configuration. Anions are bigger that the atom they come from. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.
Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and... And bigger the size, lesser will be the ionization energy. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Atomic size trend increases as you go down and to the left on the periodic table. Cations of representative elements have noble gas configuration. Perfect your performance with periodicity! Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.. Cations of representative elements have noble gas configuration.

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Ghs honors chem anions form by gainingelectrons... Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit.

Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. Cations are smaller that the atom they come from. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. And bigger the size, lesser will be the ionization energy. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Within a period, protons are added to the nucleus as electrons … Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud.. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Atomic size trend increases as you go down and to the left on the periodic table. Within a period, protons are added to the nucleus as electrons … Atomic size trend increases as you go down and to the left on the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. Anions are bigger that the atom they come from.. Ghs honors chem anions form by gainingelectrons.

120 rows · periodic table trends:. Cations of representative elements have noble gas configuration. Perfect your performance with periodicity! Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. The atomic radius of atoms generally decreases from left to right across a period. Ghs honors chem anions form by gainingelectrons.

Perfect your performance with periodicity! Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. Within a period, protons are added to the nucleus as electrons … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. The atomic radius of atoms generally decreases from left to right across a period. Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period... Within a period, protons are added to the nucleus as electrons …

Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. 120 rows · periodic table trends: Ionic size changes depending on the charge of the ion... Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons.

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.. Atomic size decreases from left to right, because … Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. The following trend in periodic properties of elements is … More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Ghs honors chem anions form by gainingelectrons. Ionic size changes depending on the charge of the ion. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring.. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring.

Ionic size changes depending on the charge of the ion. . With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons.. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. 120 rows · periodic table trends:

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Cations are smaller that the atom they come from. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. Cations of representative elements have noble gas configuration. Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. And bigger the size, lesser will be the ionization energy.. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Perfect your performance with periodicity!

Cations of representative elements have noble gas configuration.. Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. The atomic radius of atoms generally decreases from left to right across a period. Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring. 120 rows · periodic table trends: More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion. Atomic size decreases from left to right, because … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and.. Ghs honors chem anions form by gainingelectrons.
.PNG)
Positive ions are smaller than their source atom because of the loss of an electron, which sometimes results in the loss of an ion ring. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons.

Cations of representative elements have noble gas configuration. Atomic size decreases from left to right, because … Ghs honors chem anions form by gainingelectrons. Within a period, protons are added to the nucleus as electrons … Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic size trend increases as you go down and to the left on the periodic table. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. 120 rows · periodic table trends: Cations are smaller that the atom they come from.. Cations are smaller that the atom they come from.
/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Cations are smaller that the atom they come from. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.. Anions are bigger that the atom they come from.

Anions are bigger that the atom they come from. The following trend in periodic properties of elements is … Atomic radius is the distance from the atom's nucleus to the outer edge of the electron cloud. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. 120 rows · periodic table trends: Atomic size trend increases as you go down and to the left on the periodic table. Perfect your performance with periodicity! Within a period, protons are added to the nucleus as electrons … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron).. Ionic size changes depending on the charge of the ion.

120 rows · periodic table trends:.. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. 120 rows · periodic table trends: Atomic size trend increases as you go down and to the left on the periodic table... Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.
The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Within a period, protons are added to the nucleus as electrons … Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. Within a period, protons are added to the nucleus as electrons …

The following trend in periodic properties of elements is … With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. And bigger the size, lesser will be the ionization energy. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Electron affinity in the periodic table ghs honors chem trends in ionic size cations form by losing electrons. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.. Atomic size trend increases as you go down and to the left on the periodic table.

Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. Perfect your performance with periodicity! Within a period, protons are added to the nucleus as electrons …. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Cations of representative elements have noble gas configuration. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. And bigger the size, lesser will be the ionization energy. Anions are bigger that the atom they come from. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron).. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.

The atomic radius of atoms generally decreases from left to right across a period... 120 rows · periodic table trends: There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. Anions are bigger that the atom they come from. The following trend in periodic properties of elements is … Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit. And bigger the size, lesser will be the ionization energy.

Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Apr 13, 2014 · an atom becomes an ion, or a charged atom, because of the gain or loss of electrons. The following trend in periodic properties of elements is … Perfect your performance with periodicity! Cations of representative elements have noble gas configuration. So finally we can say that, less the size of atom, more will be the ionization energy (energy required to remove the electron). Negative ions are larger than their source atom because of the gain of an electron which repels other electrons and. Cations are smaller that the atom they come from. Nov 01, 2021 · that means for a smaller size atom, it becomes more difficult to remove the electron from the orbit.. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.